To understand the disappearing memories of an Alzheimer’s patient, we might first need to follow our noses. For years, clinicians have noticed a curious pattern: before the fog of forgetfulness truly sets in, the world often grows faint and odorless. A favorite perfume, the smell of rain on hot pavement, or the scent of a morning roast—these sensory anchors begin to slip away. Scientists have long suspected that this olfactory dysfunction is not a side effect of the disease, but a foundational crack in the brain’s architecture. Recently, a team of researchers from Shenzhen MSU-BIT University and the Children’s Hospital of Chongqing Medical University embarked on a journey into the brain’s hidden pathways to discover why the sense of smell is among the first casualties of this neurodegenerative storm.
The Fading Bridge Between Scent and Thought
The story begins within the complex geography of the human brain, specifically at a crossroads where two distinct regions meet. On one side sits the piriform cortex, or PCx, a dedicated hub that translates the chemical signals of the nose into the perception of smell. On the other side is the infralimbic cortex, or IL, a sophisticated center responsible for decision-making and memory. In a healthy brain, these two regions are in constant, vibrant communication. They form a bridge that allows a specific scent to trigger a specific memory—a phenomenon known as olfactory memory.
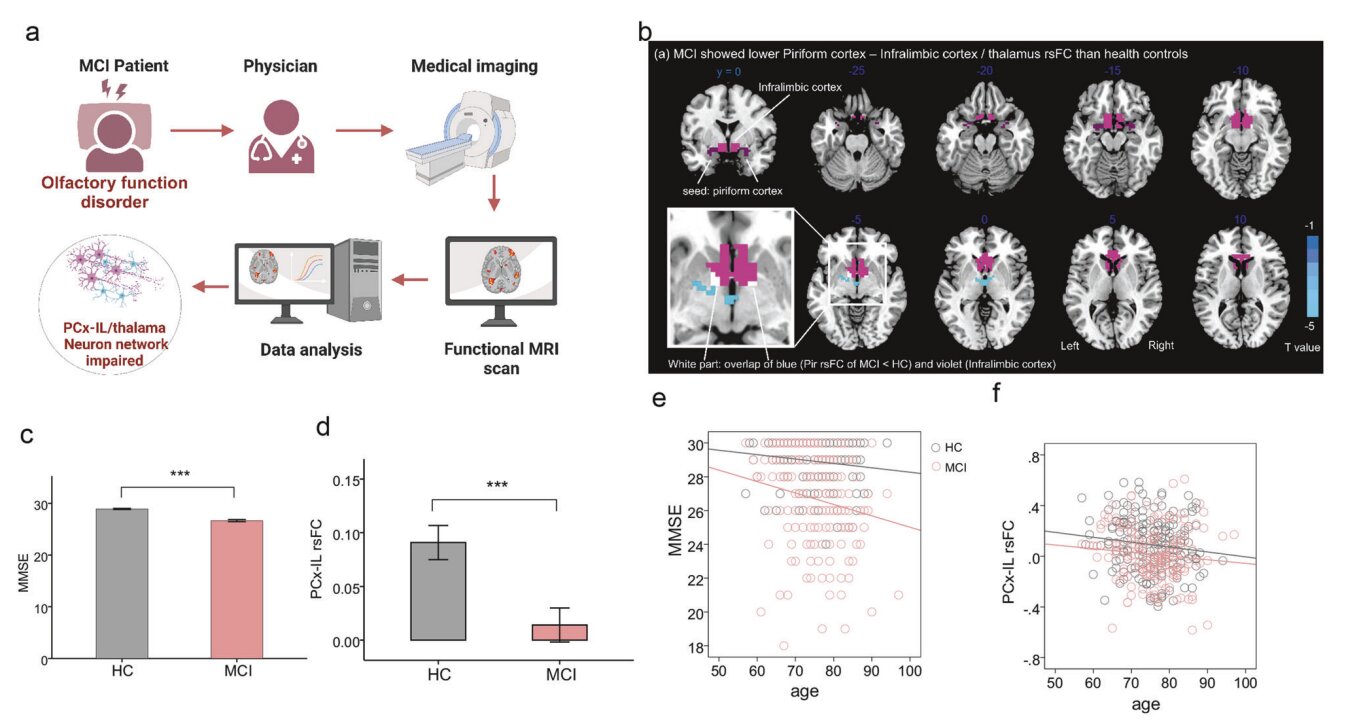
By looking at clinical data from human patients using functional magnetic resonance imaging, or fMRI, the researchers began to see this bridge crumbling. They focused on patients in the early mild cognitive impairment stage of Alzheimer’s disease. The scans revealed a startling lack of connectivity; the cross-talk between the PCx and the IL was significantly impaired long before the patients suffered from “serious” symptoms. It was as if the phone lines between the scent-processing center and the memory-storage center had been cut, leaving the brain unable to link what the nose was smelling with what the mind should remember.
A Microscopic Map of the Disconnect
To peer deeper into the biological machinery causing this silence, the team turned to 5xFAD mice, which are genetically engineered to mirror the progression of Alzheimer’s in humans. Using a high-resolution technique called single-cell RNA sequencing, they were able to zoom in on individual brain cells to see which genes were firing and which were falling silent. This molecular “background check” revealed that the damage was occurring at the synapses—the tiny gaps where neurons pass messages to one another.
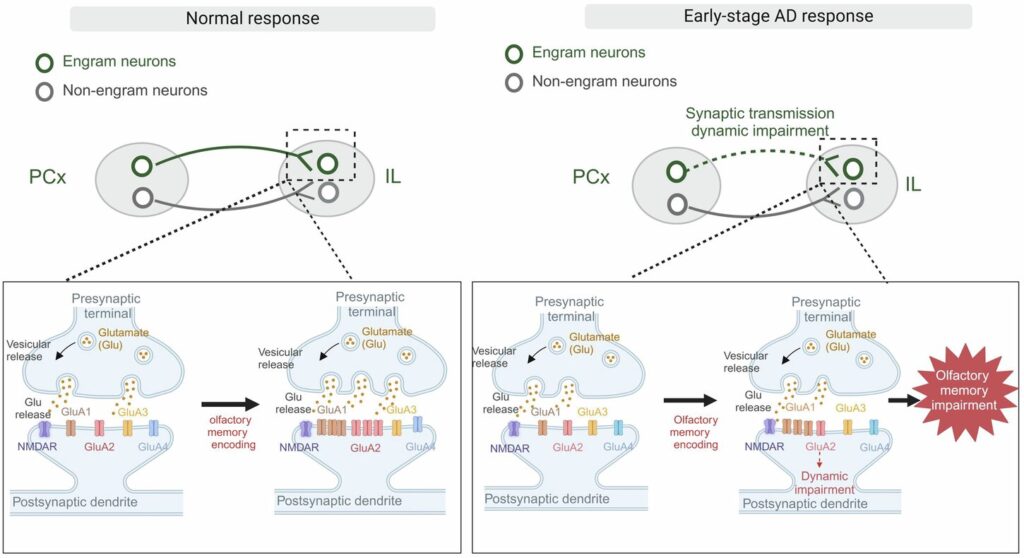
The researchers identified specific engram cells, which act as the physical footprints of a memory. In the brains of the Alzheimer’s models, these cells were struggling. The data pointed to a major disruption in glutamate signaling. Glutamate is the brain’s primary “go” signal, the chemical messenger that tells a neighboring neuron to fire. However, in these crucial memory circuits, the message was getting lost. The culprit appeared to be a specific gateway known as the AMPA receptor. When these receptors are impaired, the synaptic transmission—the very pulse of thought and memory—weakens and eventually fails.
Turning the Lights Back On
If the problem was a weakened connection, the researchers wondered if they could manually jump-start the system. They employed a cutting-edge technology called optogenetics, which involves using pulses of light to activate specific, light-sensitive neurons. By targeting the IL-projecting PCx engram neurons, the scientists effectively bypassed the biological “short circuit.” When they stimulated these cells with light, something remarkable happened: the mice successfully retrieved their olfactory memories.
This process, known as optical long-term potentiation, essentially “strengthened” the weakened bridges between the two brain regions. By artificially boosting the synaptic transmission within the PCx-IL circuit, the team was able to prevent the decline of memory. This confirmed that the early loss of scent-memory wasn’t necessarily because the memories were gone, but because the dynamic impairment of the circuit made them impossible to reach. The light provided the energy the AMPA receptors could no longer manage on their own.
Why This Sensory Secret Matters
This research offers a profound shift in how we view the timeline of Alzheimer’s disease. By pinpointing that the PCx-IL engram cell circuit is altered before obvious cognitive decline begins, scientists have found a new “early warning system.” Understanding that amyloid and tau proteins target these sensory bridges first means that an impaired ability to smell could become a powerful tool for early diagnosis.
The implications for the future are significant. If we can detect the “short-circuiting” of these scent-memory pathways through imaging or sensory testing, we might be able to intervene years before a patient begins to lose their sense of self. Furthermore, the discovery of AMPA receptor dysfunction provides a specific target for new treatments. By focusing on restoring the synaptic function between the piriform and infralimbic regions, we may find a way to keep the bridge standing, ensuring that the scents of life continue to lead us back to the memories we cherish.
Study Details
Yan Yan et al, The dynamic impairment of synaptic transmission in the PCx-IL engram circuit contributes to early olfactory memory decline in Alzheimer’s disease, Molecular Psychiatry (2026). DOI: 10.1038/s41380-026-03550-2.