Imagine a world where the complex building blocks of our medicine are created not in massive, polluting chemical plants, but through a process as clean and elegant as a leaf performing photosynthesis. For decades, the pharmaceutical industry has been trapped in a difficult balancing act. To create the highly specific molecules required for modern medicine, chemists have had to choose between two imperfect tools. On one hand, they have precise catalysts that work with surgical accuracy but are “disposable,” meaning they are used once and then thrown away, creating immense waste. On the other, they have reusable catalysts that stick around longer but often lack the finesse required for delicate chemical architecture.
A breakthrough from a research team at KAIST, led by Professor Sang Woo Han, has finally bridged this gap. By blending the strengths of two very different worlds, they have created a “living” chemical system that breathes air and drinks light to manufacture the essential ingredients of life-saving drugs. This new technology doesn’t just make the process faster; it reimagines the very nature of chemical production as a self-sustaining cycle.
The Secret Marriage of Solid and Liquid
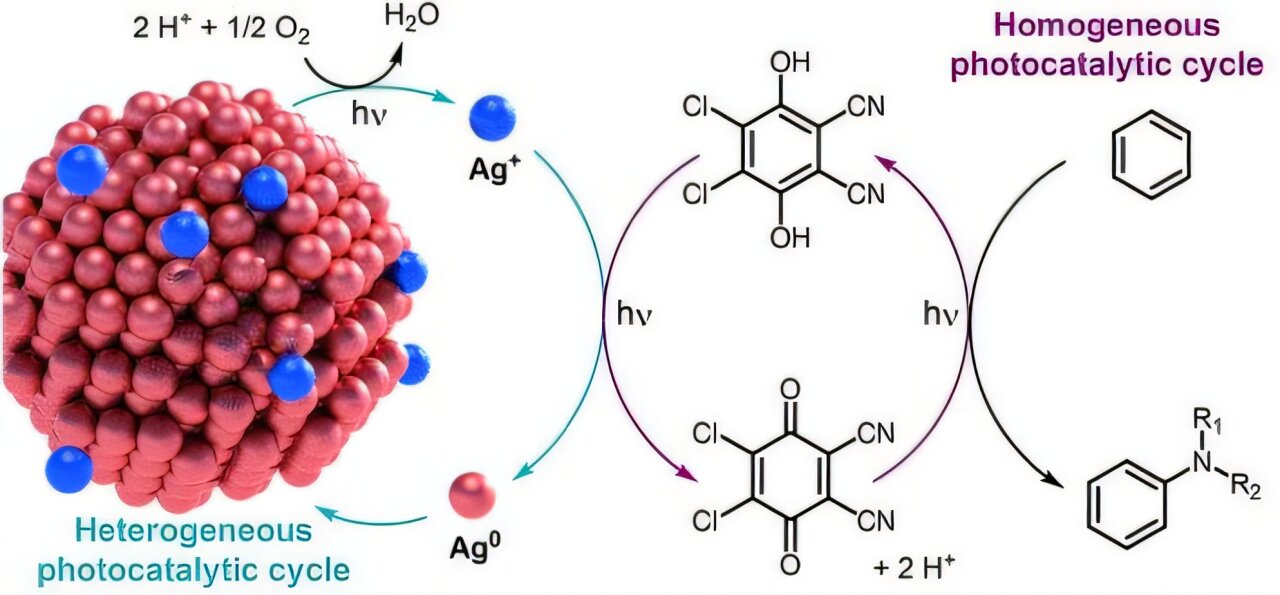
At the heart of this innovation is a sophisticated partnership between two materials that usually don’t play well together. The first is a silver-based catalyst, referred to as an Ag-based catalyst, which exists in a solid state. The second is an organic photocatalyst known as DDQ, a substance designed to spring into action the moment it absorbs a photon of light.
In the past, trying to get these two to cooperate was like trying to run a race where the runners kept tripping over one another. Their interactions were often unstable, leading to a drop in performance that made combined systems impractical. To solve this, the KAIST team introduced a mediator: lithium salt, or LiClO₄. This salt acts as a stabilizer, regulating how the silver and the DDQ interact. By managing these chemical handshakes, the researchers significantly boosted the stability and lifespan of the entire system, allowing the two catalysts to work in a synchronized harmony that was previously impossible.
A Self-Sustaining Loop of Light and Air
The most remarkable feature of this system is how it handles the problem of waste. In traditional organic photocatalysis, once a catalyst does its job, it often becomes “spent.” To get it back into a state where it can work again, scientists usually have to pump in additional chemical reagents—more “fuel” for the engine—which increases both the cost and the environmental footprint of the reaction. Sometimes, these reactions try to use oxygen from the air to reset, but the process is often too slow to be useful.
The research team bypassed this hurdle by designing a cyclic catalytic system. Instead of viewing the leftovers of a reaction as waste, they engineered a method where the byproducts generated during the chemical transformation are used to restore the catalyst. This is where the sunlight and air come in. The sunlight provides the initial spark of energy to get the reaction moving, and the oxygen in the air helps sustain the cycle, acting as the ultimate “recharging” agent.
As the reaction progresses, the catalyst continuously “recharges” itself. It moves from an active state to a used state and back again, looping indefinitely without the need for a constant infusion of fresh chemicals. Because the system relies on the atmosphere to keep the loop closed, the only byproduct left behind is water. This transforms a traditionally “dirty” industrial process into one that is exceptionally clean.
Harvesting Medicine from the Sun
To prove that this wasn’t just a theoretical success, the team put their “loop” to the test by creating amines. These are key raw materials used in a vast array of pharmaceutical ingredients. In a demonstration of pure efficiency, the researchers successfully synthesized these high-value substances using nothing but sunlight and air as the primary drivers.
This experiment proved that the inorganic photochemical loop system—a process where a metal-based catalyst reacts under light and returns to its original state—could be successfully integrated into precise organic synthesis. It showed that the “disposable” nature of high-precision chemistry is no longer a mandatory trade-off. We can now have the precision of a scalpel with the endurance of a permanent tool.
Why This Scientific Leap Matters
The implications of this research extend far beyond the laboratory walls. The chemical industry is one of the largest contributors to global carbon emissions and environmental pollution. By eliminating the need for additional chemical reagents and reducing waste to simple water, this technology offers a “green” pathway for the future of manufacturing.
It represents a dramatic reduction in the carbon footprint of the chemical industry. Because the system is powered by sunlight and maintained by air, it lowers the production speed and cost of pharmaceutical ingredients, potentially making essential medicines more affordable and accessible. As Professor Sang Woo Han noted, this is the first time such a system has been successfully applied to high-level organic synthesis, proving that we can produce the most complex compounds in the most environmentally friendly way possible. It is a vision of a future where human health and the health of the planet are no longer at odds.
Study Details
Jin Wook Baek et al, Merger of Heterogeneous and Homogeneous Photocatalysis for Arene C–H Amination, Journal of the American Chemical Society (2026). DOI: 10.1021/jacs.5c20824