The memory of the COVID-19 pandemic lingers like a shadow over our collective consciousness. For years, the world watched as an invisible enemy spread across borders, overwhelmed healthcare systems, and reshaped daily life. Vaccines and antivirals eventually arrived, but not before millions of lives were lost.
That crisis revealed a hard truth: when a new virus emerges, humanity has few defenses in the crucial early months. Unlike bacterial infections—where doctors can rely on broad-spectrum antibiotics while identifying the specific culprit—viral infections have no universal counterpart. Treatments for viruses are narrow and highly specific, effective only against a handful of related pathogens.
This gap leaves us dangerously vulnerable. A new virus can emerge at any time, and for years, the best the world can do is wait—wait for vaccines, wait for targeted antivirals, wait while lives are lost.
But now, researchers at the Nanoscience Initiative at the Advanced Science Research Center at the CUNY Graduate Center (CUNY ASRC) have made a discovery that could change that story forever. Their work, recently published in Science Advances, offers a groundbreaking path toward the development of the world’s first broad-spectrum antiviral (BSA)—a medicine that could be deployed immediately against a wide range of deadly viruses, including those we have yet to encounter.
The Global Health Challenge
To understand the magnitude of this breakthrough, we must first grasp the challenge. Viruses are shape-shifters. They evolve quickly, and each one is different enough that a drug designed for one rarely works against another. This is why antivirals are typically designed to target very specific viral families. Tamiflu, for example, works against influenza but is useless against Ebola. Antivirals for HIV do nothing against coronaviruses.
This narrowness has left humanity exposed. “This lack of treatments can leave populations vulnerable for years, while vaccines and therapeutics are being developed,” explained Adam Braunschweig, principal investigator of the study, professor with the Nanoscience Initiative at CUNY ASRC, and a Chemistry and Biochemistry professor at Hunter College.
Braunschweig and his team asked a bold question: instead of chasing viruses one by one, could we find a common vulnerability—a molecular Achilles’ heel—that runs across many viral families?
A Hidden Target on Viral Surfaces
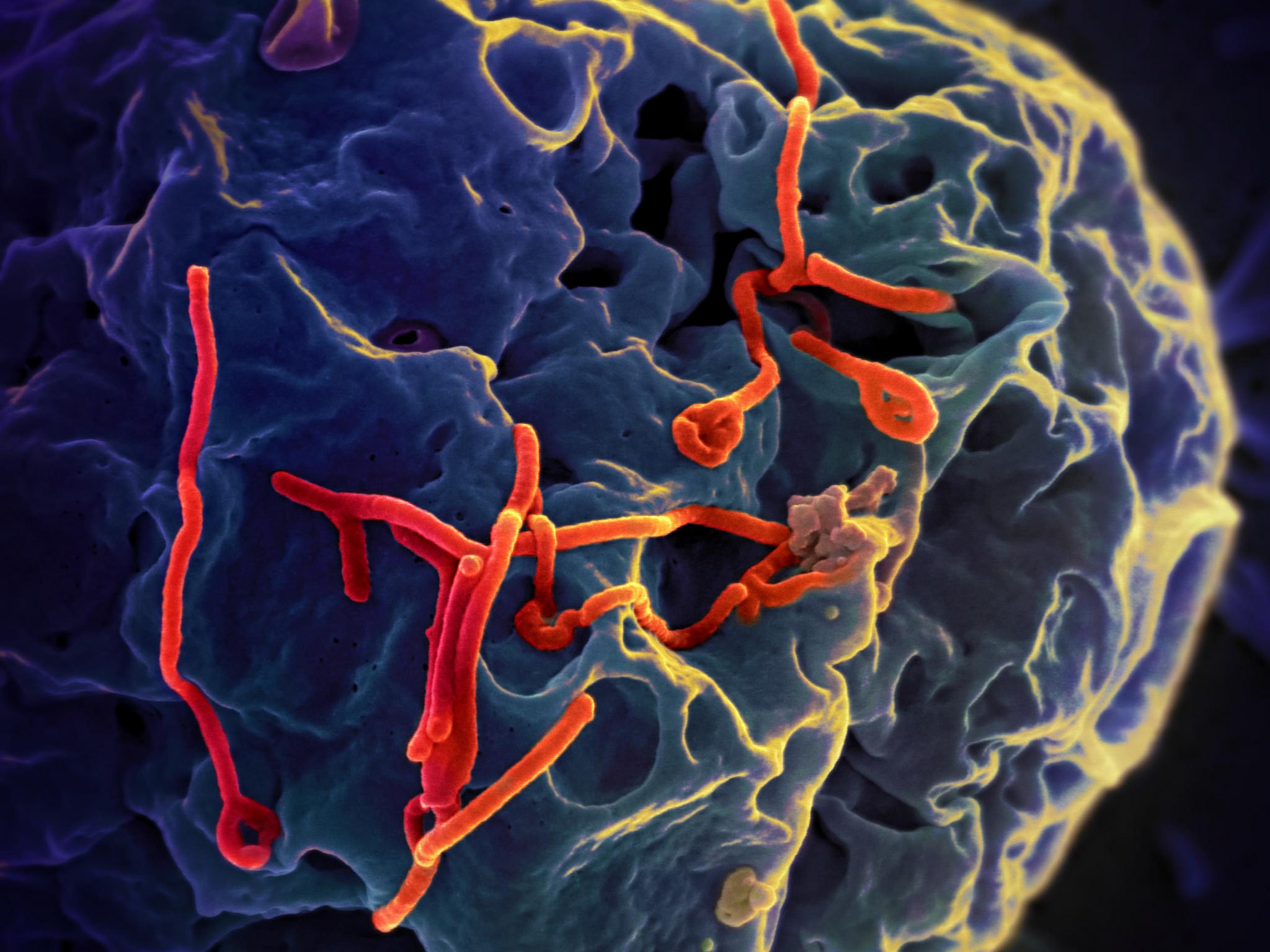
The answer lay on the very surface of viruses themselves. Many viruses, including some of the deadliest known to humankind, are coated with sugar molecules called glycans. These viral envelope glycans are structurally conserved, meaning they appear again and again across unrelated viruses—from Ebola to coronaviruses to Nipah.
Until now, this feature had been largely ignored as a drug target. But Braunschweig’s team recognized its potential. If they could design molecules that specifically bound to these glycans, they might have a weapon that could lock onto a wide range of viruses at once, neutralizing them before they could infect cells.
To test this daring idea, the researchers turned to synthetic carbohydrate receptors (SCRs)—small molecules engineered to recognize and bind to specific sugar structures.
The Breakthrough Discovery
The team screened 57 different SCRs, each designed to interact with viral glycans. Out of this library, they identified four lead compounds that showed remarkable activity.
These molecules were able to block infection from seven different viruses across five unrelated viral families. This included some of the most feared pathogens on Earth: Ebola, Marburg, Nipah, Hendra, SARS-CoV-1, and SARS-CoV-2.
The implications are staggering. For the first time, scientists had found small molecules that could stop a diverse range of lethal viruses using a single mechanism.
But the most dramatic test came in animals. When mice infected with SARS-CoV-2 were treated with one of the lead SCR compounds, 90 percent survived. In the untreated control group, none did. Further analysis confirmed that the compounds worked exactly as predicted—by binding to viral glycans and blocking their ability to infect cells.
“This is the kind of antiviral tool the world urgently needs,” Braunschweig said. “If a new virus emerges tomorrow, we currently have nothing to deploy. These compounds offer the potential to be that first line of defense.”
A New Mechanism, New Possibilities
The discovery goes beyond just infectious disease. Because glycans also play critical roles in cancer biology and immune disorders, the team’s novel approach may open doors to entirely new therapeutic strategies across medicine. A drug that targets glycans could, in theory, be a multipurpose weapon against a spectrum of diseases that until now have required very different treatments.
What makes this especially exciting is the mechanism of action. Traditional antivirals target viral enzymes or replication machinery, but viruses mutate quickly and can often develop resistance. By targeting conserved glycans—a feature that viruses rely on and cannot easily change—these SCRs may prove harder for viruses to evade.
A Glimpse of the Future
The journey from laboratory discovery to medicine is long and complex. The next steps for Braunschweig’s team involve advancing the most promising compounds into clinical trials, where safety and effectiveness in humans can be rigorously tested.
If successful, the world may one day have a stockpile of broad-spectrum antivirals, ready to be deployed at the first sign of a new outbreak. Imagine a future where, instead of waiting years for vaccines or therapies, doctors could immediately prescribe a drug that buys precious time—time to contain outbreaks, save lives, and prevent pandemics from spiraling out of control.
That vision is no longer science fiction. It is within reach.
Hope in the Face of Uncertainty
The story of science is often the story of persistence against impossible odds. From the discovery of antibiotics to the mapping of DNA, progress has always depended on daring minds who asked, “What if?”
This breakthrough in antiviral research is one of those moments. It is a testament not only to human ingenuity but to our collective determination to be better prepared for whatever nature throws our way.
Viruses will not stop evolving. New threats will come, as they always have. But with discoveries like this, humanity is no longer destined to always play catch-up.
The future may finally hold a defense as adaptable, as broad, and as resilient as the viral enemies we face. And that could change everything.
More information: Broad-spectrum Synthetic Carbohydrate Receptors (SCRs) Inhibit Viral Entry Across Multiple Virus Families, Science Advances (2025). DOI: 10.1126/sciadv.ady3554