Deep within the microscopic landscape of the yeast Saccharomyces cerevisiae, a silent biological clock is ticking. For decades, geneticists have treated this humble model organism as a window into the most profound mysteries of human life, seeking to understand how cells manage to divide over and over again without falling apart. The secret lies at the tips of our chromosomes, in the protective caps known as telomeres. These repetitive DNA sequences act like the plastic tips on shoelaces, preventing our genetic code from fraying or degrading. Yet, with every cycle of life, these caps wear down.
The Clock That Counts Our Days
Every time a cell divides to create a new version of itself, its telomeres undergo a process of gradual shortening. This is not merely a byproduct of biology; it is a fundamental mechanism of aging. When these protective barriers become too short, the cell reaches a state called cellular senescence, where it essentially retires and stops dividing. In most of our body’s somatic cells, this clock is relentless, contributing to the natural aging of our tissues. However, life requires certain cells to be immortal—or at least more resilient. During embryonic development, and within our stem cells, germ cells, and specific immune cells, the shortening must be counteracted to ensure the body can grow and repair itself indefinitely.
This is where a remarkable enzyme named telomerase enters the story. Telomerase acts as a molecular construction crew, lengthening the telomeres and allowing cells to undergo extended rounds of division while preserving chromosomal integrity. While this enzyme is the hero of development, it has a dark side. In approximately 90% of cancer cells, telomerase is abnormally reactivated. This “hijacking” allows tumors to achieve cellular immortality, dividing without end. To understand the mechanisms behind both aging and cancer, scientists knew they had to see this enzyme not just as a concept, but as a physical machine.
A Frozen Glimpse into the Invisible
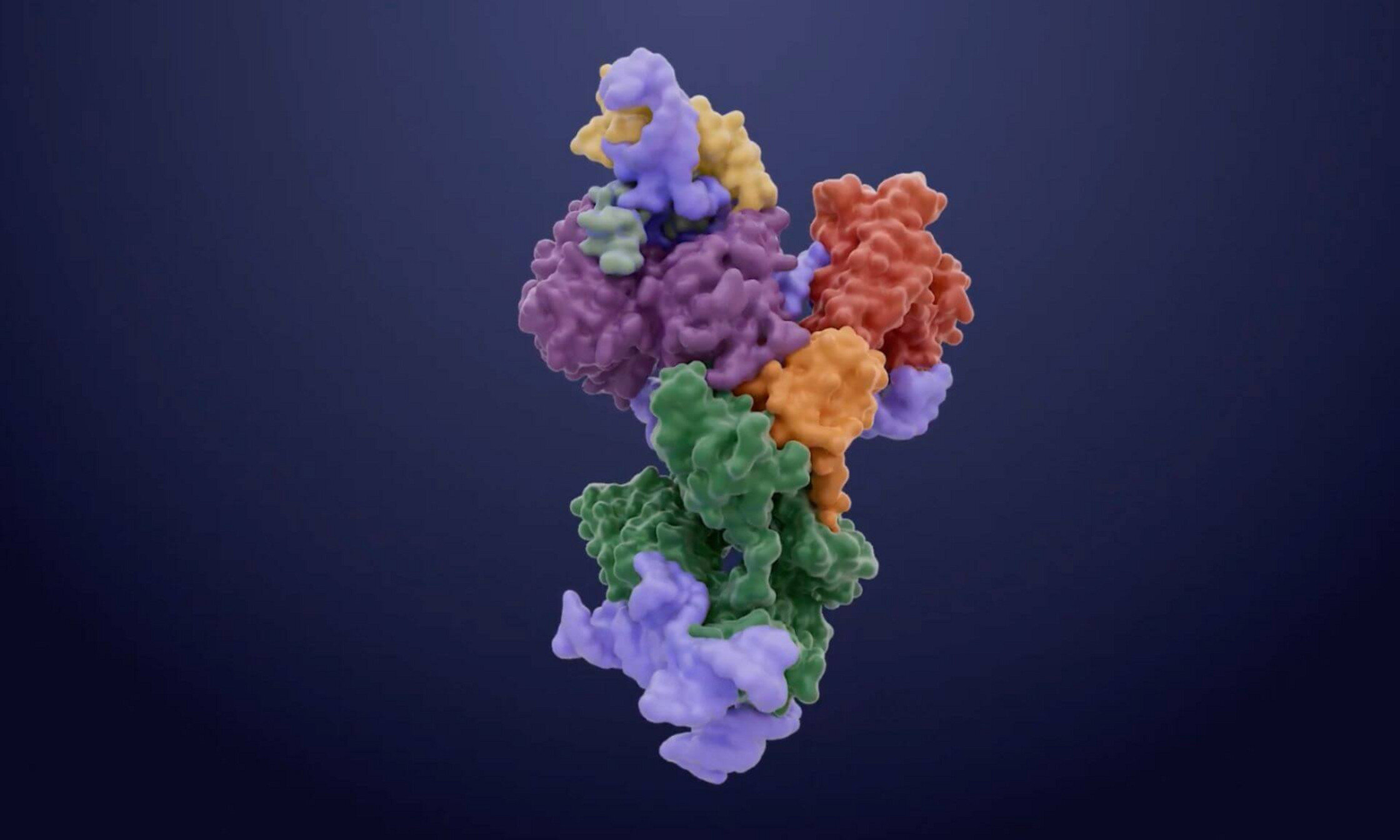
For years, the exact shape of telomerase remained a blurry enigma. Traditional methods like X-ray crystallography or nuclear magnetic resonance struggled to capture the complexity of such a large molecular machine. To break through this barrier, an international collaboration led by Pascal Chartrand of the Université de Montréal, alongside researchers from the Université de Sherbrooke and the MRC Laboratory of Molecular Biology, turned to a revolutionary technology: cryogenic electron microscopy.
The process began with the painstaking purification of the telomerase complex. Once isolated, the team used extreme cold to flash-freeze the enzyme, trapping it in a thin layer of vitreous ice. This “cryogenic” state preserved the delicate architecture of the proteins and RNA in their natural configuration. Using the electron microscope, the team captured millions of individual images from every conceivable angle. High-powered software then stitched these snapshots together to reconstruct a three-dimensional model at near-atomic resolution. The level of detail was staggering, reaching a scale of just a few angstroms—where one angstrom is a mere ten-millionth of a millimeter. For the first time, the internal gears of the yeast telomerase were laid bare.
Uncovering the Hidden Finger of Life
As the three-dimensional map materialized, the researchers spotted something that had never been seen before in this specific enzyme: a zinc finger. In the world of molecular biology, a zinc finger is a recurring structural pattern typically found in proteins that need to grip onto DNA or RNA. Its presence in telomerase was an “unexpected feature” that changed the team’s understanding of how the enzyme operates.
Pascal Chartrand and his colleagues hypothesized that this tiny, finger-like protrusion was responsible for binding a specific portion of the telomerase RNA, effectively stimulating the enzyme’s activity. To prove it, they performed a delicate genetic surgery, mutating the zinc finger pattern. The result was definitive: the enzyme’s activity vanished almost entirely. This confirmed that the zinc finger was not just a decorative ornament but a vital ignition switch for the machine.
The Architect Holding the Machine Together
The structural map revealed another crucial player in this microscopic drama: a protein called Est3. While scientists knew Est3 was involved in the process, its exact role had remained a mystery. The high-resolution imagery revealed that Est3 serves as a molecular scaffold. It acts as the physical bridge that links the various components of the telomerase complex together. Without this scaffold to ensure structural integrity, the entire enzyme would lose its shape and its ability to function. Chartrand noted that this protein is absolutely essential for the enzyme to remain active within the cell, providing the framework upon which the work of DNA extension is built.
Why This Microscopic Map Matters
The discovery of these “conserved” protein interactions—features that remain similar across different species—provides a new roadmap for the future of medicine. By identifying the specific elements that are indispensable to telomerase function, such as the zinc finger and the Est3 scaffold, scientists have identified new “targets” for future therapies.
The implications are dual-pronged. In the fight against cancer, finding ways to disable these specific components could provide a method to strip tumors of their immortality, forcing them to succumb to the natural aging process of senescence. Conversely, for patients suffering from telomeropathies—rare disorders where telomeres fail prematurely—or for those dealing with the debilitating effects of tissue aging, these insights could lead to treatments that modulate or enhance telomerase activity. By understanding the three-dimensional geometry of how our chromosomes are protected, we move one step closer to mastering the biological clock that governs our health and our lifespan.
Study Details
Hongmiao Hu et al, Cryo–electron microscopy structure of the budding yeast telomerase holoenzyme, Science (2026). DOI: 10.1126/science.adz5344