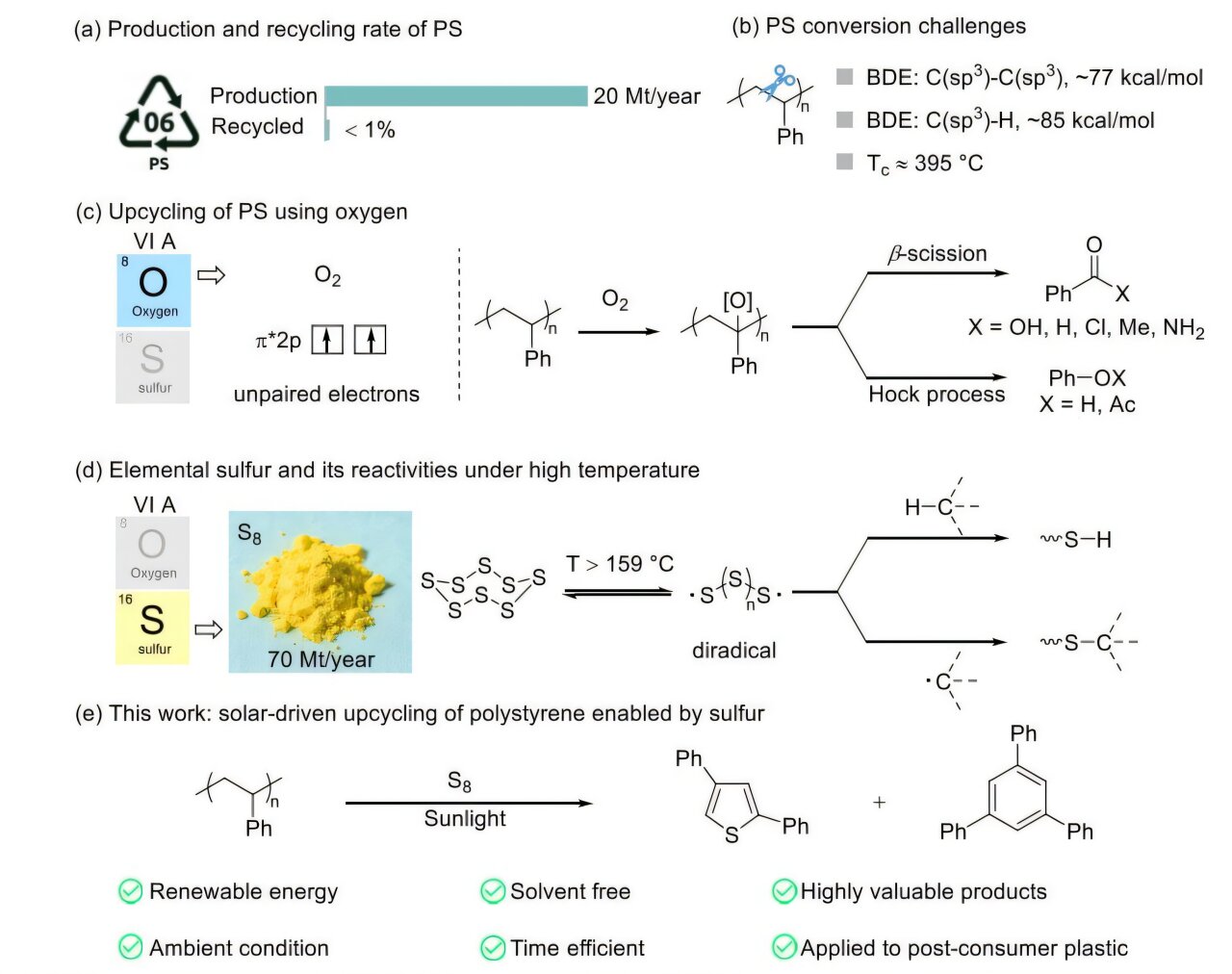
The world is currently drowning in a sea of white foam and clear plastic. From the disposable spoons tossed away after a single lunch to the yogurt containers and packing foam that shield our electronics, polystyrene is everywhere. It is prized by industry for being incredibly cheap and durable, but that same durability is a curse for the planet. Every year, we produce over 20 million tons of this material, yet a staggering 1% of it is successfully recycled. The rest sits in landfills, stubborn and unchanging, because its chemically inert nature makes it nearly impossible to break down without massive amounts of energy or expensive chemicals.
But in a quiet revolution of chemistry, a team of researchers in China has looked at this mountain of waste and seen a goldmine. They have pioneered a way to turn one of the planet’s most persistent pollutants into high-value components for the semiconductor industry, and they are doing it by harnessing two of the most overlooked materials on Earth: surplus sulfur and raw sunlight.
The Ghost of the Oil Industry Meets the Ghost of the Lunchroom
The secret to this transformation lies in an unlikely partnership. For decades, the process of purifying crude oil and other industrial activities has produced a massive byproduct: elemental sulfur (S₈). This bright yellow substance is generated in a surplus of millions of tons, often sitting forgotten in giant, open-air stockpiles for years because the world simply has more of it than it knows what to do with.
The researchers realized that this “waste” sulfur possessed unique electronic properties that could solve the “waste” plastic problem. In their experiment, they gathered common polystyrene waste—the kind of plastic that usually ends up in the gut of a sea bird or buried in a trench—and mixed it directly with this elemental sulfur. There were no expensive lab reagents or toxic solvents involved; just two types of industrial leftovers brought together to see if they could create something new.
A Two Minute Dance Under a Focused Beam
To trigger the reaction, the team didn’t plug in a heater or fire up a furnace. Instead, they turned to the sky. They placed the mixture of plastic and sulfur under a concave mirror, a simple tool designed to catch and concentrate sunrays. This is where the elemental sulfur reveals its true magic. It acts as a photothermal agent, meaning it is exceptionally good at absorbing sunlight and converting that light directly into intense heat.
Under the focused glare of the sun, the temperature of the mixture spiked to approximately 300 °C in moments. Within just two minutes, the solid white plastic and yellow sulfur melted and morphed into a reddish-black liquid. The reaction happened in ambient conditions, meaning it didn’t require a vacuum or a pressurized chamber. It was a rapid, solar-powered metamorphosis.
Through computer simulations and lab tests, the scientists discovered exactly what was happening at the molecular level. The heat from the sun caused the sulfur to form reactive sulfur radicals. These aggressive little molecules acted like chemical hooks, reaching out to pull hydrogen atoms away from the polystyrene chains. This “kick-start” broke the plastic’s stubborn chemical bonds and allowed the atoms to rearrange themselves into entirely different, more complex shapes.
Crafting the Building Blocks of the Digital Future
When the reddish-black liquid cooled and was processed, the researchers found they hadn’t just destroyed the plastic; they had upcycled it into two specific, high-value molecules. The first, 2,4-diphenylthiophene, was recovered with a 34% yield. This molecule is a prize for the tech world, as it can be transformed into materials used in optoelectronics and high-performance semiconductors.
The second molecule, 1,3,5-triphenylbenzene, was recovered with a 16% yield. This molecule is characterized by a unique, rigid planar structure. Because it is flat and sturdy at a molecular level, it serves as an ideal building block for creating advanced functional materials. By using the sun to stitch together waste plastic and waste sulfur, the team created the very materials needed to build the next generation of computers and electronic sensors.
Why This Sun-Drenched Discovery Matters
This research represents a fundamental shift in how we view the “trash” of the modern world. For years, polystyrene recycling has been a failing endeavor because the energy required to break it down often cost more than the resulting material was worth. By using clean energy from the sun and a surplus byproduct like sulfur, this method removes the high cost and high carbon footprint of traditional recycling.
It solves three problems at once: it provides a destination for the millions of tons of sulfur sitting in stockpiles, it offers a rapid, solvent-free way to keep plastic waste out of landfills, and it creates a domestic supply of chemicals for the semiconductor industry. This is the essence of polymer upcycling. Instead of just melting a plastic spoon to make a lower-quality plastic spoon, we are now able to transform that spoon into the “brains” of our digital devices. As this process scales, it offers a practical, elegant path toward a circular economy where nothing is truly wasted, and even the most stubborn pollutants can be reborn through the power of a few minutes of concentrated sunlight.
Yong Liu et al, Solar-Driven Upcycling of Polystyrene Enabled by Elemental Sulfur, Journal of the American Chemical Society (2026). DOI: 10.1021/jacs.6c01318