The human brain is often described as the final frontier of medical science, a vast territory where memories are stored and identities are forged. But what if the secrets to one of the most devastating brain disorders are actually hidden much lower, in the winding channels of the digestive tract? For years, Alzheimer’s disease has been viewed almost exclusively as a condition of the mind, defined by a slow, tragic fading of the self. Yet, a growing body of evidence suggests that the story of this disease may begin in the gut, long before the first name is forgotten.
A Quiet Storm in the Gut
Before the fog of memory loss sets in, many people who will later be diagnosed with Alzheimer’s disease experience a different kind of distress. They suffer from gastrointestinal dysmotility, most commonly manifesting as chronic constipation. While these symptoms were once dismissed as unrelated side effects of aging, researchers at Nantes University in France began to wonder if the gut was actually a primary battlefield.
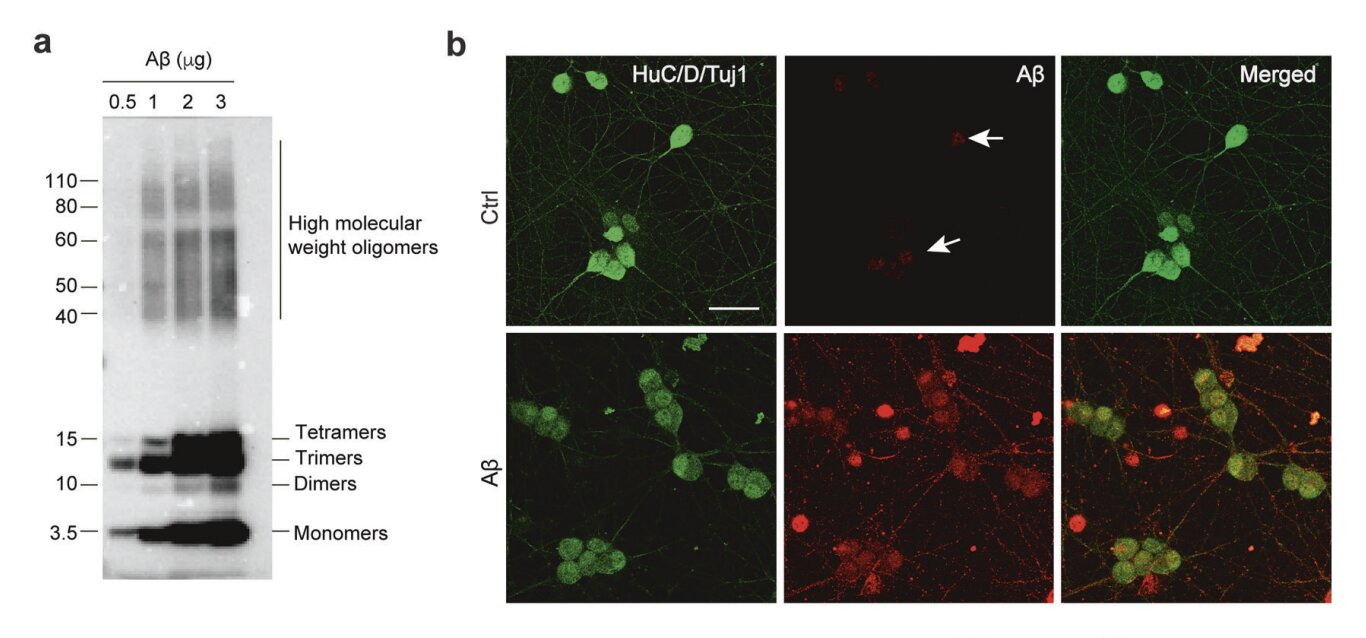
The traditional understanding of the disease centers on a protein called amyloid-β, or Aβ. In a healthy brain, this protein is managed effectively, but in those with the disease, it clumps together to form amyloid plaques. These sticky deposits act like roadblocks, disrupting the vital communication between neurons and eventually leading to the death of brain cells. The French team, led by Rodrigue Brossaud and Thibauld Oullier, hypothesized that this same toxic buildup wasn’t just happening in the skull—it was simultaneously occurring in the enteric nervous system, the intricate web of nerves that governs our digestion.
The Hidden Map of the Second Brain
To test this theory, the researchers turned to a specific group of subjects: SAMP8 mice. These mice are unique because they naturally develop symptoms that mirror the progression of human Alzheimer’s. By examining these animals, the team could look at the very earliest stages of the disease, catching the pathology in the act before it caused irreversible damage.
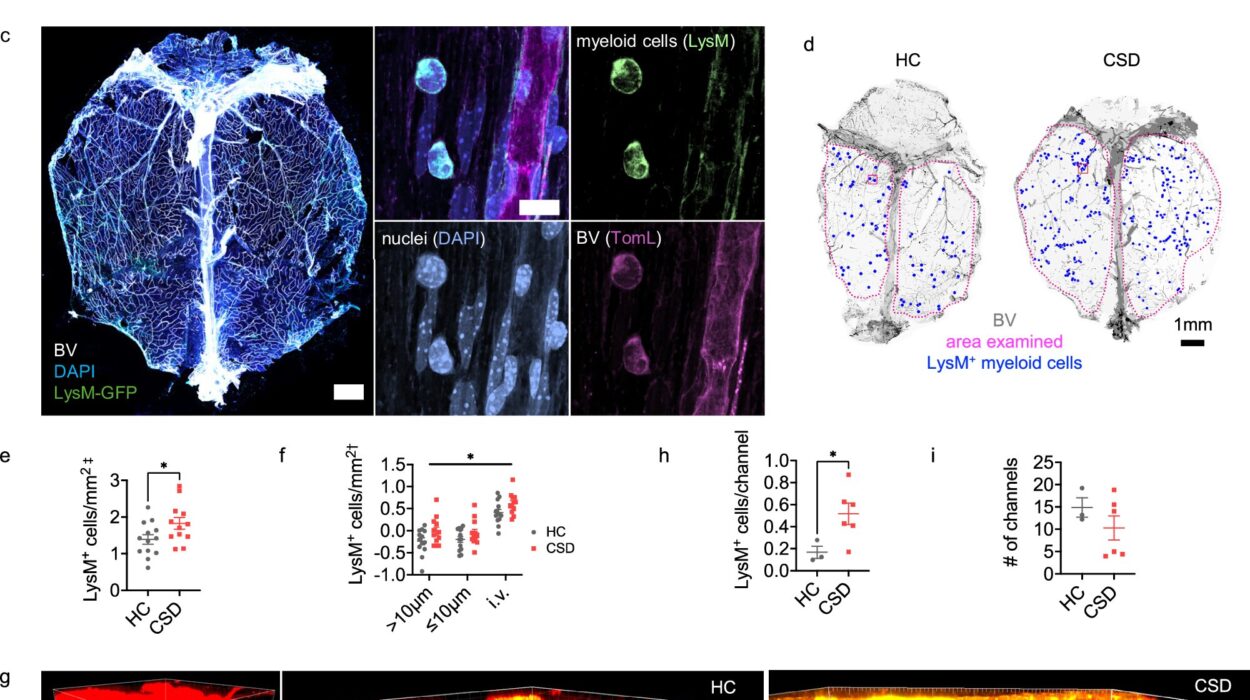
The investigation revealed a striking parallel between the gut and the brain. In the intestines of these mice, the researchers found a significant rise in amyloid pathology. They discovered that an enzyme called BACE1, which is responsible for producing the toxic Aβ, was working overtime. Conversely, an enzyme called neprilysin, which usually acts as a molecular janitor to clear away the protein, was in short supply. This imbalance created a perfect storm for amyloidosis, a state where the protein accumulates unchecked.
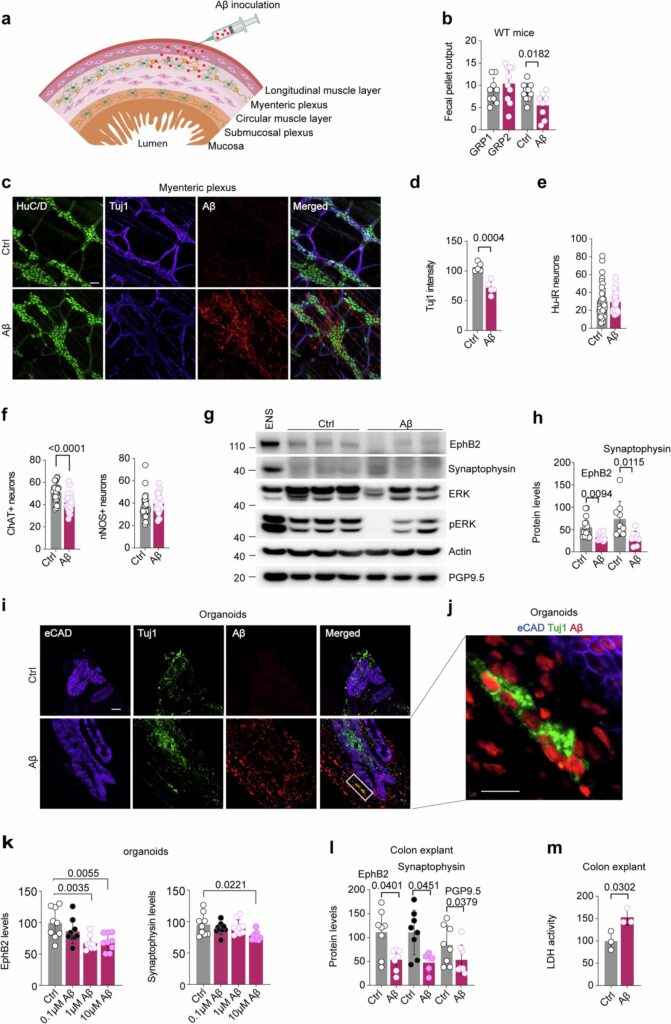
This buildup wasn’t just sitting there; it was actively dismantling the “wiring” of the gut. The researchers observed a degradation of key synaptic-associated proteins known as EphB2 and synaptophysin. Without these proteins, the connections in the enteric nervous system began to fray. The mice didn’t just have a brain disease; they had a system-wide failure of connectivity that started in their bellies.
From Petri Dishes to Living Systems
The team didn’t stop with mice. To ensure their findings had relevance for humans, they explored the effects of Aβ on human colon tissue and organoids—miniature, lab-grown versions of human organs derived from stem cells. The results were hauntingly consistent. When exposed to the toxic protein, these human models showed the same “lesions” and broken connections seen in the mice.
To prove that the gut could actually drive the disease, the scientists performed a daring experiment. They delivered Aβ directly into the colons of healthy, wild-type mice. Soon after, these healthy mice began to exhibit the same gastrointestinal symptoms and nerve damage seen in the Alzheimer’s models. This confirmed a chilling possibility: the presence of these proteins in the gut is enough to trigger the physical hallmarks of the disease.
A Microscopic Ally in the Fiber
In the search for a solution, the researchers looked toward a humble molecule called butyrate. This is a short-chain fatty acid produced naturally by “good” bacteria in the gut when they break down dietary fiber. It is a byproduct of a healthy microbiome, and the team wondered if it could act as a shield against the encroaching amyloid tide.
The results of the butyrate treatment were remarkable. When the SAMP8 mice were given the molecule, the production of Aβ plummeted. The butyrate didn’t just stop the protein from forming; it protected the existing nerve connections and significantly lowered inflammation throughout the body.
The most stunning revelation, however, was the effect on the mind. The mice treated with butyrate did not develop the memory deficits that typically plague their kind. By stabilizing the gut and reducing the toxic load of Aβ in the plasma and digestive tract, the researchers had effectively shielded the brain from the hallmark symptoms of dementia.
Why This Connection Changes Everything
This research marks a pivotal shift in how we conceptualize neurodegenerative disease. It suggests that the brain and the gut are not two separate entities, but part of a continuous loop where the health of one intimately dictates the fate of the other. By identifying that amyloid-β accumulation begins in the gut and travels to the brain—possibly through gut-to-brain transmission—scientists have found a new window for intervention.
If we can detect the rise of enzymes like BACE1 or the decline of neprilysin in the digestive tract years before memory loss begins, we might be able to treat the disease in its infancy. The discovery that a simple molecule like butyrate can block Aβ accumulation and prevent gut dysfunction offers a glimmer of hope for future therapies. While these findings must still be validated in human clinical trials, they provide a roadmap for preventing the “irreversible damage” that has long made Alzheimer’s so fearsome. We are learning that to save the mind, we may first need to listen to what the gut has been trying to tell us all along.
Study Details
Rodrigue Brossaud et al, The short-chain fatty acid butyrate prevents gut-brain amyloid-β pathology and neuroinflammation in an Alzheimer mouse model, Molecular Psychiatry (2026). DOI: 10.1038/s41380-026-03522-6.