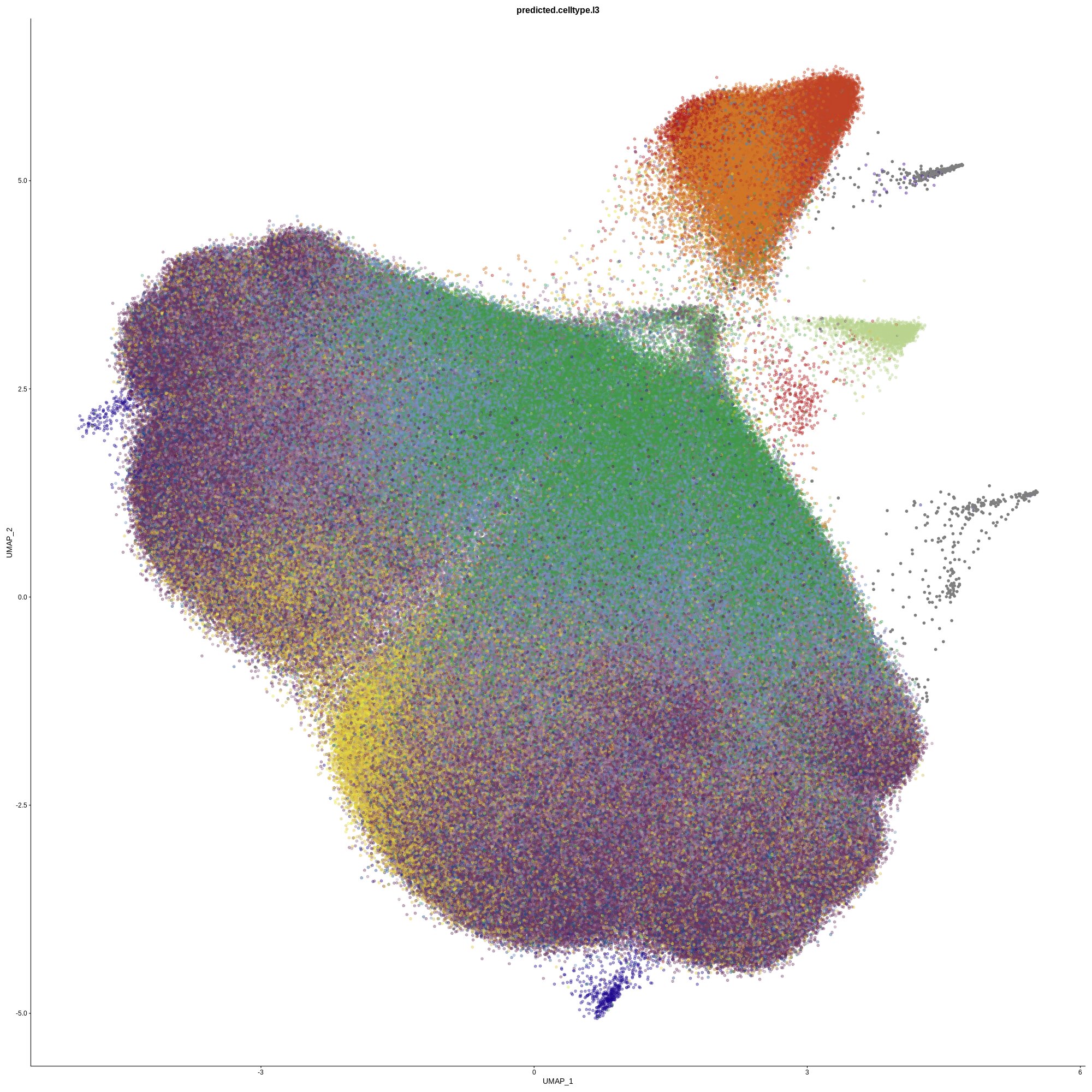
New research from the Garvan Institute and UNSW Sydney has identified more than 1,000 genetic control switches that behave differently in female and male immune cells, driving higher inflammatory activity in females. By analyzing over 1.25 million immune cells from nearly 1,000 healthy people at single-cell resolution, the study offers a clearer genetic explanation for why diseases like lupus affect up to nine women for every one man. The findings strengthen calls for sex-specific approaches in medical research and treatment.
Autoimmune diseases have long carried a striking imbalance: women are far more likely than men to develop conditions where the immune system mistakenly attacks the body. For decades, scientists have recognized the pattern, but the deeper biological explanation has remained incomplete.
Now, a large-scale study ahead of World Lupus Day on May 10 is helping to clarify why that imbalance exists. Researchers found that the immune systems of women and men are not only functionally different, but genetically regulated in different ways—at a level of detail that older methods could not detect.
The study was led by scientists at the Garvan Institute of Medical Research and UNSW Sydney and published in The American Journal of Human Genetics.
A clearer explanation for a long-standing medical mystery
Diseases like lupus can affect up to nine women for every one man, but the reasons behind this dramatic sex bias have been difficult to pin down. While hormones and other biological influences have been considered, researchers have lacked detailed evidence showing exactly how genetic regulation differs between the sexes inside immune cells.
This new research suggests that a major part of the answer lies in how genes are controlled in women versus men.
The team discovered over 1,000 genetic switches that operate differently depending on sex, shaping immune activity and pushing female immune cells toward stronger inflammatory responses.
Dr. Seyhan Yazar, the study’s first author from Garvan, said the findings highlight why immune research must account for sex differences instead of treating male biology as the default.
“Our findings show that the immune system needs to be studied with sex in mind,” Yazar said, noting that many studies still fail to separate results by sex, which can limit understanding and skew treatment development.
Why single-cell analysis changed everything
For years, immune differences between men and women were mostly studied using bulk blood analysis. That approach measures average gene activity across a mixed population of cells.
The problem is that blood contains many different immune cell types, each with its own behavior. When everything is blended together, important differences inside specific cell populations can disappear.
This study used newer single-cell technologies, allowing researchers to examine immune activity at an unprecedented level of detail. Instead of averaging signals, the team could see what was happening in individual immune cells.
The researchers sequenced more than 1.25 million peripheral blood mononuclear cells from nearly 1,000 healthy individuals. All participants came from the OneK1K cohort, a major Australian project focused on mapping how genetics affects immune cells across large populations.
This scale made it possible to detect subtle but widespread differences that older studies would have missed.
Male and female immune cells showed distinct profiles
Once the team analyzed the data, a clear pattern emerged: male and female immune systems were not simply different in degree, but different in composition and genetic focus.
Males had higher proportions of monocytes, immune cells that act as first responders. Their genetic activity was more focused on basic cellular upkeep, including maintenance and protein-building functions.
Females, in contrast, had higher levels of B cells and regulatory T cells, and their genetic activity was strongly biased toward inflammatory pathways.
Dr. Sara Ballouz, co-senior author and Senior Lecturer at UNSW, explained that this immune setup comes with both advantages and risks.
A more reactive immune system can improve defense against viral infections, but it also raises the likelihood of immune overreaction.
Ballouz described this as a biological trade-off: females may be better primed to fight infections, but that heightened readiness increases the chances of immune “friendly fire,” where the body becomes its own target.
The genetic switches that control immune intensity
The researchers went deeper by investigating what they described as sex-specific genetic switches—variations that influence how strongly genes are turned on or off in immune cells.
These switches are known as expression quantitative trait loci, and they function like volume controls for gene expression.
Because the team analyzed immune cells individually, they were able to detect sex-specific genetic regulation that bulk studies could not uncover. This led to one of the study’s most striking findings: these sex-linked regulatory differences were not concentrated where many scientists expected them to be.
A surprise finding: most differences were not on sex chromosomes
Many researchers have assumed that the largest immune differences between women and men would be driven mainly by the X and Y chromosomes.
But the study found that sex-specific genetic switches were far less common on sex chromosomes than expected.
Instead, the vast majority were located on autosomes, the non-sex chromosomes shared by both males and females.
In total, the team identified over 1,000 sex-specific genetic switches on autosomes. This suggests that immune differences between sexes may be rooted far more broadly in the genome than previously assumed.
Direct links to lupus-related genes
Beyond mapping immune activity, the researchers also found evidence connecting these sex-specific genetic controls to real autoimmune conditions.
They identified specific variants affecting the female-biased expression of two genes associated with systemic lupus erythematosus.
This may help explain why lupus occurs at dramatically higher rates in women, aligning with the widely observed statistic of nine times higher prevalence in women compared to men.
While the researchers emphasized that genetics is only one piece of the puzzle—alongside influences such as hormones—these genetic differences may establish a baseline immune state that shapes long-term disease risk.
Ballouz described this as a new layer of understanding in human immunity, showing that sex differences exist at the level of genetic regulation itself.
Rethinking treatment in autoimmune disease
The study also raises an important clinical issue: if female and male immune systems are fundamentally regulated differently, then autoimmune disease may not present the same way in both sexes.
That matters because many autoimmune treatments are still broadly designed around generalized immune suppression, rather than targeting the specific pathways driving disease in different individuals.
The researchers suggest that understanding sex-specific inflammatory pathways could eventually support more precise therapies, shifting away from broad immunosuppressants toward treatments tailored to how a person’s immune system is genetically wired.
Yazar said the findings strengthen the case that autoimmune disease should not be approached with a universal model.
“Our findings add strong evidence that female and male autoimmune diseases may not be the same,” Yazar said, emphasizing that current clinical practice often relies on one-size-fits-all management.
Why This Matters
This research provides some of the clearest evidence yet that sex-based differences in immunity are not just hormonal or superficial—they are built into genetic regulation across the immune system. By identifying over 1,000 sex-specific genetic switches and linking some directly to lupus-associated genes, the study offers a more concrete explanation for why autoimmune diseases disproportionately affect women.
It also reinforces a major message for modern medicine: treating men and women as biologically identical in research can lead to incomplete understanding and potentially biased treatment strategies. As scientists push toward precision medicine, studies like this highlight that sex is not a minor variable—it may be a core factor shaping disease risk, immune behavior, and how well treatments work.
Study Details
The impact of sex on the immune system explored at the single-cell level, The American Journal of Human Genetics (2026). DOI: 10.1016/j.ajhg.2026.04.003. www.cell.com/ajhg/fulltext/S0002-9297(26)00153-9