The golden years are often shadowed by a quiet, creeping fear: the loss of memory. For generations, we have accepted the slowing of the mind as a universal tax on longevity, an inevitable decay of the brain itself. Yet, if we look closer at the world around us, the rule seems to have curious exceptions. Some individuals remain strikingly sharp at 100 years old, while others begin to feel the fog of forgetfulness as early as middle age. This discrepancy suggests that aging might not be a simple clock winding down, but a complex conversation between our organs—one that we are only just beginning to overhear.
The Secret Language Within Our Walls
While it seems logical to blame a fading memory on the aging of the hippocampus or the degeneration of neurons within the skull, new research published in Nature suggests the culprit may reside much further down. Our bodies possess two ways of sensing the world. Most of us are familiar with exteroception—the five senses of sight, hearing, taste, smell, and touch that allow us to navigate our external environment. But there is a silent, internal counterpart known as interoception. This is the subconscious process by which the brain monitors the state of our internal organs to keep the body in balance.
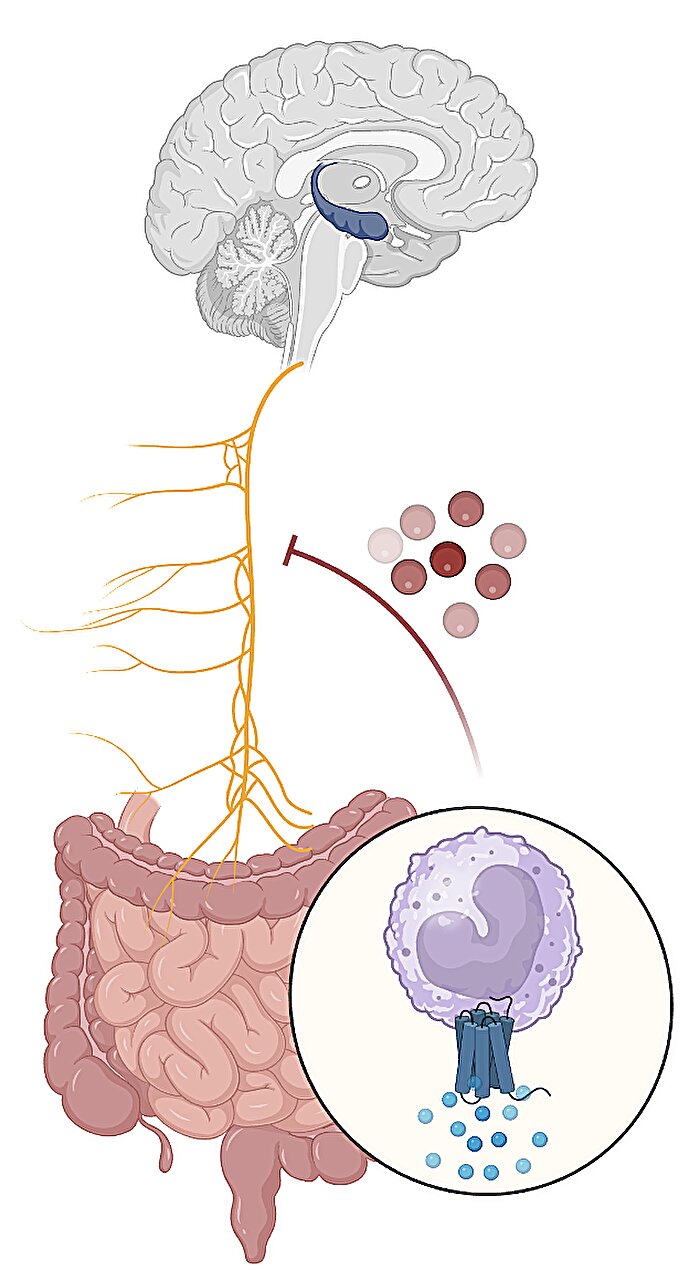
The primary highway for this internal dialogue is the vagus nerve. It is a massive neural connector that links the heart, lungs, liver, and intestines directly to the brain. Scientists have discovered that this intestine-to-brain signaling pathway acts as a protective shield against cognitive decline. When this pathway is healthy, it helps maintain youthful memory function. However, just as our eyes grow dim and our ears lose their edge, our interoceptive senses also decline with age. The internal “hearing” of the brain starts to fail, not because the brain is broken, but because the message from the gut is being muffled.
A Hostile Takeover in the Microscopic Dark
To understand why this internal signal weakens, researchers turned their attention to the gut microbiome—the vast ecosystem of microbes inhabiting our digestive tracts. As we age, the composition of this microbial city shifts; some populations dwindle while others thrive, changing the very chemistry of our literal “gut feelings.” To test the power of these microbes, scientists introduced the microbiome from old mice into young ones. Almost immediately, the young mice began to struggle with memory tasks, performing as poorly as their elderly counterparts.
The most startling revelation came when looking at germ-free mice, which are raised without any microbiome at all. These mice showed a significantly slowed rate of cognitive decline compared to normal mice. It appeared that a byproduct of the aging microbiome was actively driving the process of memory loss. By narrowing down the search, researchers identified a specific bacterial suspect: Parabacteroides goldsteinii. As this microbe becomes more abundant with age, it floods the system with molecules known as medium-chain fatty acids (MCFAs).
The Chain Reaction of Forgetfulness
The accumulation of these MCFAs triggers a biological domino effect. These fatty acids activate myeloid immune cells residing in the gut, which in turn begin to pump out inflammatory signaling molecules. One molecule in particular, a cytokine called IL-1β, acts like static on a radio line. It impairs the function of the vagal sensory neurons, effectively “blunting” the activity of the gut-brain highway.
The researchers traced this path with clinical precision: the bacteria produce the MCFAs, which stir the immune cells, which release the IL-1β, which then numbs the vagus nerve. Without the clear, protective signals traveling from the gut to the hippocampus, the brain’s ability to form and retrieve memories begins to wither. It is a discovery that redefines “brain aging” not as an isolated neurological failure, but as a systemic breakdown caused by a noisy, inflamed gut.
Rewiring the Connection to the Past
If the cause of memory loss lies in a chemical buildup in the gut, then perhaps the cure does as well. The researchers explored several ways to break the cycle of decline. One method involved using antibiotics to deplete the microbiome, which successfully restored youthful cognitive function in mice. While effective, this isn’t a long-term solution for health. A more surgical approach involved using a bacteriophage—a virus that specifically targets and limits the activity of P. goldsteinii. This intervention lowered MCFA levels and, remarkably, improved the memories of the mice.
Even more promising for future human applications is the idea of bypassing the bacteria altogether and speaking directly to the vagus nerve. By treating mice with the gut hormone CCK or with GLP-1 receptor agonists—drugs similar to those used in modern treatments like Ozempic—the researchers were able to stimulate the nerve and reverse memory deficits. This suggests that the “eyeglasses” for our internal senses might already exist in the form of existing pharmaceuticals.
Why the Gut Holds the Key to the Mind
This research fundamentally shifts our understanding of the aging process. It proves that cognitive decline is not an untouchable, locked-away mystery of the skull, but a process that can be influenced, controlled, and even reversed by managing the body’s peripheral organs. If what we call “brain aging” is actually a result of vagus nerve dysfunction driven by gut inflammation, we gain a massive advantage. The gut and the immune system are far easier to manipulate and treat than the delicate tissues of the brain itself.
While these studies were conducted in mice, the bridge to human health is already being built. Patients who use implanted devices for vagus nerve stimulation to treat epilepsy or stroke have already reported unexpected improvements in their memory and focus. This suggests that our own internal pathways are just as sensitive to these signals. By learning to tune the conversation between our gut and our brain, we may one day be able to treat the fog of old age not as an inevitable ending, but as a technical glitch that can be fixed, allowing us to stay present and connected for as long as we live.
Study Details
Christoph Thaiss, Intestinal interoceptive dysfunction drives age-associated cognitive decline, Nature (2026). DOI: 10.1038/s41586-026-10191-6. www.nature.com/articles/s41586-026-10191-6