Every time we scrape a knee, battle an infection, or recover from inflammation, an invisible drama unfolds inside our bodies. At the cellular level, healing is not simply a matter of patching things up—it’s a carefully choreographed dance of destruction and rebirth. Cells must clear away damaged parts, reset their machinery, and sometimes even revert to a younger, more flexible state in order to regrow healthy tissue.
For decades, scientists have known about processes like programmed cell death, which helps remove damaged cells, and about paligenosis, a reprogramming mechanism that allows mature cells to roll back into a stem cell-like state to fuel tissue regeneration. But now, researchers at Washington University School of Medicine in St. Louis and the Baylor College of Medicine have uncovered something entirely new: a dramatic cellular purge they’ve named cathartocytosis—from Greek roots meaning “cellular cleansing.”
This process, first described in a study published in Cell Reports, could reshape our understanding of how cells heal—and why, under certain conditions, healing goes awry.
A Cellular Shortcut With a Cost
Healing requires speed. When a tissue is damaged, cells must shift focus from their day jobs—digesting food, contracting muscles, transmitting signals—to emergency repair work. But here’s the problem: the very machinery that allows a cell to perform its normal duties can get in the way of becoming a regenerative powerhouse.
“After an injury, the cell’s job is to repair that injury. But the cell’s mature machinery for doing its normal job gets in the way,” explained Dr. Jeffrey W. Brown, first author of the study. “So, this cellular cleanse is a quick way of getting rid of that machinery so it can rapidly become a small, primitive cell capable of proliferating and repairing the injury.”
In other words, cathartocytosis is a shortcut. Instead of slowly breaking down and recycling old components in a controlled manner inside cellular compartments called lysosomes, the cell simply ejects them. Dr. Brown compared the process to vomiting—messy, but effective.
This rapid decluttering allows the cell to quickly reset into a flexible, stem cell-like state. But like any shortcut, there are risks. The ejected cellular waste doesn’t just disappear—it lingers in the surrounding tissue, where it can spark inflammation or, under chronic conditions, even feed cancer development.
Discovery in the Stomach
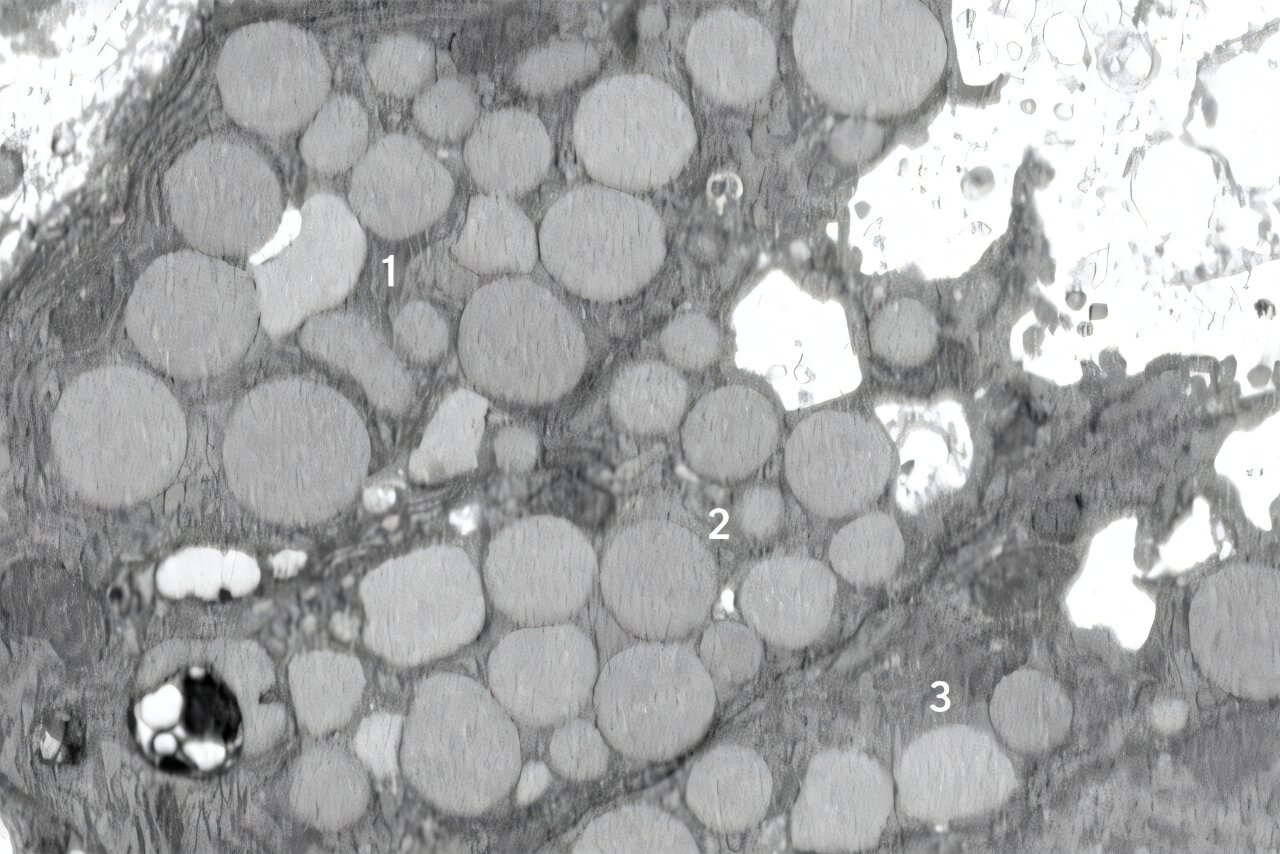
The breakthrough came while studying stomach injuries in mice. Researchers were investigating paligenosis, the well-documented process in which cells rewind into a more primitive state to fuel regeneration. Initially, they believed all cellular decluttering during this transition happened quietly inside lysosomes. But then they began noticing something unusual: debris outside the cells.
At first, the scientists dismissed it as an accident—a cellular “spill.” But the more they looked, the more consistent the phenomenon became. When stomach cells across the tissue simultaneously reverted to stem cell-like states, they all began ejecting internal components at once.
This wasn’t an accident. It was a coordinated response.
Cathartocytosis, it turned out, was not a bug in the healing system, but a feature. A rapid, dramatic purge that cells use when time is of the essence.
The Double-Edged Sword
If cathartocytosis is nature’s shortcut to healing, it also comes with hidden dangers. By expelling damaged cellular machinery into the surrounding tissue, the process creates a messy environment. Under normal circumstances, the body can clean up the debris and move forward. But in the case of chronic injury or infection, the waste piles up—and trouble begins.
Persistent cathartocytosis may fuel ongoing inflammation, keeping tissues trapped in a cycle of damage and repair. This constant cellular stress, combined with the tendency of aging cells to accumulate mutations, creates fertile ground for cancer.
Dr. Jason C. Mills, senior author of the study, emphasized the stakes: “In these gastric cells, paligenosis—reversion to a stem cell state for healing—is a risky process, especially now that we’ve identified the potentially inflammatory downsizing of cathartocytosis within it.”
In the stomach, this could explain why chronic infections with Helicobacter pylori, a bacterium known to cause ulcers, increase the risk of cancer. Cells caught in a loop of cathartocytosis may continuously shed waste and mutate, laying the foundation for malignant growth.
A Potential Biomarker for Cancer
While the discovery of cathartocytosis raises concerns, it also opens new doors for diagnosis and treatment. If the waste expelled during this process can be detected, it might serve as an early warning sign of trouble.
Dr. Brown and colleagues have already developed an antibody that binds to this expelled material, potentially offering a way to track when—and where—cathartocytosis is happening. In the future, this could become a tool for detecting precancerous states, catching disease before it fully takes hold.
“If we have a better understanding of this process, we could develop ways to help encourage the healing response and perhaps, in the context of chronic injury, block the damaged cells undergoing chronic cathartocytosis from contributing to cancer formation,” Dr. Brown explained.
A New Chapter in the Story of Healing
Cathartocytosis adds a new dimension to our understanding of how the body heals itself. It reminds us that healing is not a neat, orderly affair but a raw, sometimes messy process. Cells take risks, cut corners, and improvise in ways that can save tissue—or, under the wrong conditions, set the stage for disease.
This discovery also highlights the delicate balance nature strikes between efficiency and safety. To survive, organisms need fast healing mechanisms. But speed can come at the cost of precision, and when cells purge too recklessly or too often, the very process designed to protect life can threaten it.
Looking Ahead
The identification of cathartocytosis is only the beginning. Researchers now want to know: Does this process occur in other tissues beyond the stomach? How common is it in the body’s response to injury? Could it be harnessed to improve healing in acute injuries, while suppressed in chronic inflammatory diseases or cancer?
The answers could transform medicine, offering new ways to promote regeneration or prevent malignancy. And as with so many discoveries in science, cathartocytosis reminds us that the smallest details—microscopic expulsions inside a single cell—can carry profound implications for life, health, and survival.
In the end, cathartocytosis is more than just a new word in biology. It is a vivid example of the ingenuity of living systems, their relentless drive to heal, and the precarious balance between repair and risk that defines the biology of being alive.
More information: Jeffrey W. Brown et al, Cathartocytosis: Jettisoning of cellular material during reprogramming of differentiated cells, Cell Reports (2025). DOI: 10.1016/j.celrep.2025.116070