For centuries, the secrets of the human mind have been intertwined with the chemistry of the natural world. Deep within certain fungi, hidden in the waxy skin of the Sonoran Desert toad, and pulsing through the veins of specific botanical species, lie compounds capable of altering perception and potentially mending broken spirits. These substances, known as indolethylamines, include famous names like psilocybin, psilocin, and DMT. While they have long been part of ancient spiritual traditions, modern medicine is now looking at them through a different lens: as a revolutionary toolkit for treating depression, anxiety, PTSD, and addiction.
The challenge, however, isn’t just understanding how these molecules interact with our brains; it’s finding a way to create them that doesn’t destroy the very ecosystems where they originate. Harvesting rare toads or over-exploiting wild mushrooms creates an ecological debt that science is eager to avoid. Traditional laboratory synthesis is an option, but it is often a clunky, multi-step process that requires specific, sometimes difficult-to-handle chemical reactants and can leave behind unwanted byproducts. Seeking a more harmonious solution, a team of researchers turned to a surprising ally: a humble relative of the tobacco plant known as Nicotiana benthamiana. They didn’t just want to grow the medicine; they wanted to teach the plant how to build it from scratch.
Rewriting the Botanical Blueprint
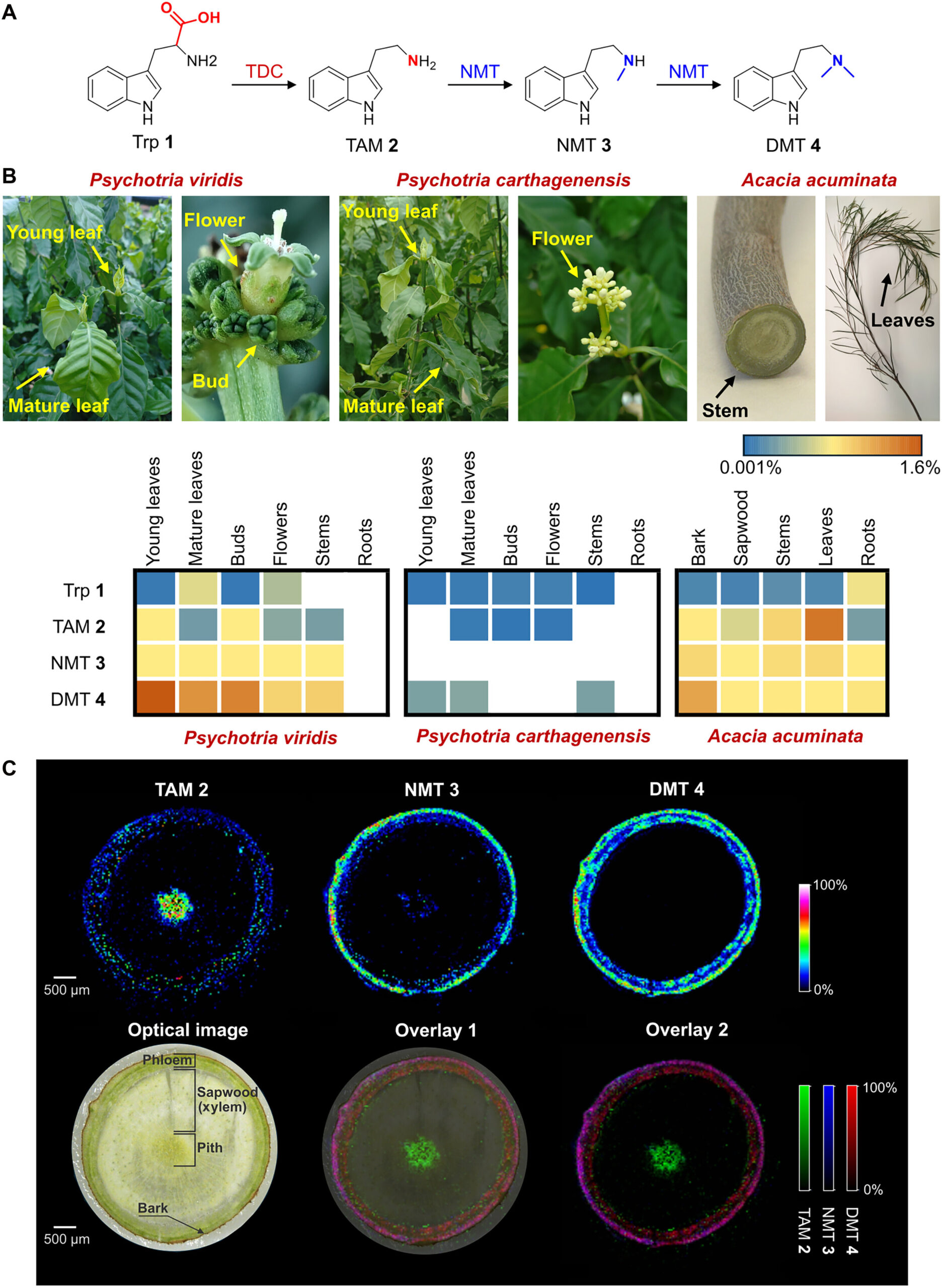
The journey began with a master class in biological cartography. To turn a plant into a miniature pharmaceutical factory, the researchers first had to map the biosynthetic pathways of five major natural psychedelics: DMT, psilocin, psilocybin, bufotenin, and 5-MeO-DMT. This required looking across the entire tree of life, identifying the specific enzymes—the biological machines—that toads, fungi, and other plants use to assemble these complex molecules.
The researchers selected Nicotiana benthamiana for this experiment because it is a fast-growing, easily cultivated plant that naturally produces high levels of tryptophan, a vital amino acid that serves as the raw building block for all indolethylamine compounds. The goal was to “plug in” the genetic instructions from other species into the tobacco plant’s own metabolic system. To do this, they used a technique called agroinfiltration. In this process, the leaves are gently injected with a bacterial suspension that carries the new genetic code, essentially giving the plant a temporary set of instructions to start producing something entirely new.
Artificial Intelligence Meets Ancient Chemistry
Simply giving a plant the instructions wasn’t enough; the researchers wanted the production to be efficient. To optimize the process, they looked toward the cutting edge of technology, employing AlphaFold3. This advanced AI model is capable of predicting the 3D structures and complex interactions of molecules with startling accuracy. By using the AI to guide their hand, the team was able to design mutant proteins that were far more effective than their natural counterparts.
In one instance, the AI suggested a rational design involving a single amino acid substitution in a specific enzyme. This tiny, calculated tweak resulted in a staggering 40-fold increase in the production of 5-MeO-DMT. This synergy between digital intelligence and biological engineering allowed the team to bypass the slow trial-and-error of evolution, creating a streamlined pathway for the plant to churn out these therapeutic compounds. The result was a true “proof of concept”: a single tobacco plant that, for the first time, contained all five natural psychedelic compounds simultaneously.
Beyond the Natural World
The researchers didn’t stop at replicating what already exists in nature. By manipulating the biosynthetic pathways, they were able to create halogenated indolethylamine analogs. These are “designer” versions of psychedelics that are not typically found in the wild. These synthetic cousins hold immense interest for researchers because small changes in molecular structure can lead to vastly different effects in the body.
For example, the study notes that certain variations, like 5-chloro-DMT and 5-fluoro-DMT, produce traditional hallucinogenic responses in animal models, while others, like 5-bromo-DMT, act as sedatives. Even more intriguing is the 5,6-dibromo version, which has shown antidepressant-like activity. By turning the tobacco plant into a programmable platform, the scientists have opened the door to a world of “structural diversification,” where they can potentially tailor-make molecules to target specific symptoms of mental health disorders without the side effects often associated with traditional drugs.
A Sustainable Horizon for Mental Health
While the current concentrations of these compounds in the engineered plants are lower than those found in their original natural sources, the achievement marks a foundational shift in how we think about drug production. This isn’t just about growing a single crop; it’s about establishing a versatile platform that can be scaled and adapted. The researchers envision a future where these pathways could be integrated into edible plants for precise microdosing applications or even adapted for microbial systems like yeast or bacteria.
This research matters because it addresses the “bottleneck” of modern psychedelic medicine. As clinical interest explodes and more drugs receive “Breakthrough Therapy” designations, the demand for high-quality, ethically sourced compounds will skyrocket. By blending the catalytic functions found across the tree of life into a single, manageable plant system, we are moving toward a future where life-saving mental health treatments are as sustainable as they are effective. It represents a bridge between the ancient power of natural medicine and the precision of modern genetic engineering, ensuring that the path to healing the human mind does not come at the cost of the planet’s biodiversity.
Study Details
Paula Berman et al, Complete biosynthesis of psychedelic tryptamines from three kingdoms in plants, Science Advances (2026). DOI: 10.1126/sciadv.aeb3034