For centuries, the human body was understood as a mysterious and intricate machine whose inner workings were shaped by invisible instructions. These instructions—encoded within DNA—determine the color of our eyes, the structure of our cells, and even our vulnerability to certain diseases. For most of modern history, scientists could only observe these genetic instructions. They could read parts of the biological script, but they could not rewrite it.
That changed with the emergence of CRISPR.
CRISPR represents one of the most transformative scientific discoveries of the twenty-first century. It offers scientists a way to edit genes with a level of precision, efficiency, and accessibility that was once unimaginable. What once sounded like science fiction—the ability to repair faulty genes, eliminate inherited diseases, and redesign biological systems—has become an active field of medical research.
The implications reach far beyond the laboratory. CRISPR technology has the potential to reshape medicine, agriculture, biotechnology, and even the future of human evolution. It carries enormous promise, but it also raises profound ethical questions about how humanity should use the power to alter life itself.
To understand CRISPR is to understand a turning point in science. It is the moment when humans moved from simply reading the genetic code to actively editing it.
The Blueprint of Life
Every living organism is built according to a molecular blueprint. This blueprint is written in DNA, a long molecule composed of chemical units called nucleotides. Arranged in precise sequences, these nucleotides form the genetic instructions that guide growth, development, and cellular function.
Inside nearly every cell of the human body lies the entire genome—a complete set of genetic instructions inherited from our parents. These instructions determine how proteins are made, how cells communicate, and how tissues function together to sustain life.
Genes are segments of DNA that contain the instructions for building specific proteins. Proteins, in turn, carry out most of the work inside cells. They control chemical reactions, provide structural support, regulate biological processes, and allow cells to respond to their environment.
When genes function correctly, they produce proteins that support healthy biological activity. But when genetic mutations occur, the instructions can become faulty. These errors may cause proteins to malfunction or fail entirely, leading to disease.
Thousands of human disorders are caused by such genetic mutations. Conditions like sickle cell disease, cystic fibrosis, and Huntington’s disease arise from changes in a single gene. Others involve complex interactions among multiple genes.
For decades, scientists dreamed of correcting these mutations directly at the genetic level. Instead of treating symptoms, they hoped to repair the root cause.
CRISPR has brought that dream within reach.
The Origins of CRISPR in Bacteria
The story of CRISPR begins not in human medicine but in bacteria. These microscopic organisms, despite their simplicity, possess surprisingly sophisticated defense systems.
Bacteria constantly face attacks from viruses known as bacteriophages. These viruses invade bacterial cells and hijack their molecular machinery to reproduce. To survive, bacteria evolved a remarkable immune strategy.
Within their DNA, bacteria store fragments of genetic material from viruses that previously infected them. These fragments serve as molecular memories of past invaders. When the same virus attacks again, the bacteria use these stored sequences to recognize and destroy the viral DNA.
This defense mechanism is known as CRISPR, an acronym for Clustered Regularly Interspaced Short Palindromic Repeats. The name refers to repeating patterns found within bacterial genomes.
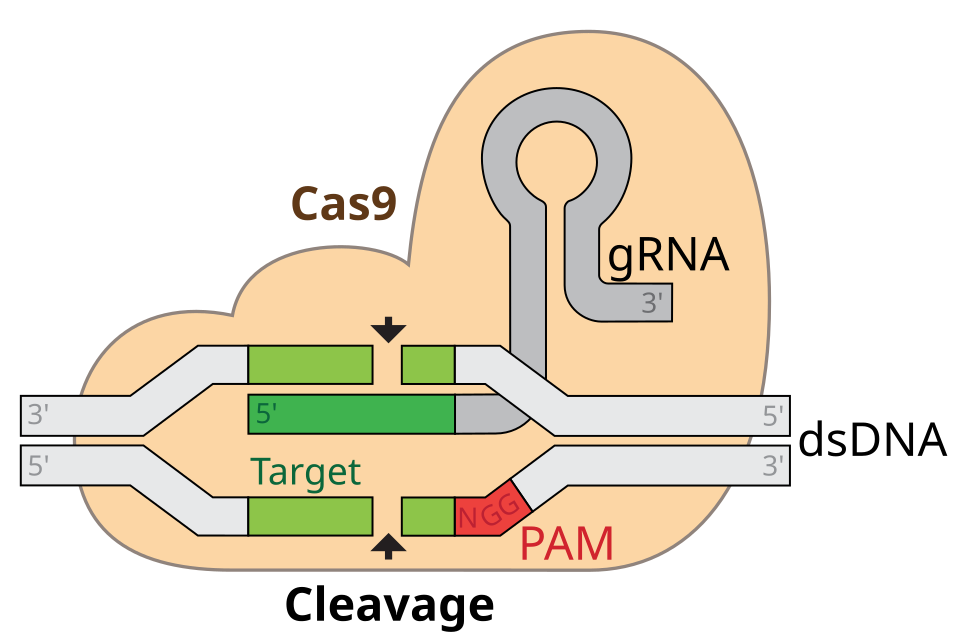
Associated with these CRISPR sequences are specialized proteins called Cas proteins. These proteins act like molecular scissors. Guided by RNA molecules that match viral DNA sequences, Cas proteins locate invading genetic material and cut it apart, preventing infection.
For bacteria, CRISPR is a powerful immune system.
For scientists, it turned out to be something even more extraordinary.
The Discovery That Changed Biology
For years, the repeating sequences in bacterial DNA puzzled researchers. Their function remained unclear until scientists began recognizing patterns linking them to viral genetic material.
Gradually, researchers realized that bacteria were storing fragments of viral DNA as part of an adaptive immune defense. This discovery revealed that microorganisms possessed a memory-based immune system—a surprising concept for organisms once thought to be biologically simple.
As scientists studied the mechanism further, they noticed something remarkable. The CRISPR system could be programmed to target specific DNA sequences with extraordinary accuracy.
This meant that if researchers could design a guide RNA that matched a chosen genetic sequence, the Cas protein could be directed to cut DNA at that exact location.
In other words, the bacterial immune system could be transformed into a gene-editing tool.
This insight sparked a scientific revolution.
By harnessing the CRISPR-Cas system, researchers suddenly had the ability to edit DNA in living cells with unprecedented precision.
How CRISPR Gene Editing Works
CRISPR gene editing relies on a simple yet powerful concept: targeted DNA cutting followed by cellular repair.
The process begins with the design of a guide RNA. This RNA molecule contains a sequence that matches the DNA region scientists want to modify. It acts like a molecular GPS, guiding the Cas enzyme to the correct location within the genome.
When the CRISPR system enters a cell, the guide RNA binds to the Cas protein. Together they move through the cell’s DNA, scanning for a sequence that matches the guide RNA.
Once the correct target is found, the Cas enzyme cuts both strands of the DNA helix.
At this moment, the cell’s natural repair mechanisms take over.
Cells cannot tolerate broken DNA, so they quickly attempt to repair the cut. Scientists can use this repair process to introduce genetic changes. If the DNA rejoins imperfectly, it may disable a gene entirely. Alternatively, researchers can provide a replacement DNA template that the cell uses during repair, allowing precise insertion or correction of genetic information.
Through this process, CRISPR allows scientists to delete genes, modify them, or insert entirely new sequences.
The technology functions like an incredibly precise editing tool for the genetic code.
Why CRISPR Is Revolutionary
Gene editing technologies existed before CRISPR, but they were often slow, expensive, and technically complex. Many earlier methods required extensive protein engineering to target specific DNA sequences.
CRISPR changed the landscape because it relies primarily on RNA guidance. Designing a new guide RNA is far easier than engineering a new protein.
This simplicity dramatically reduced the cost and complexity of gene editing. Laboratories around the world suddenly gained access to powerful genetic tools.
The result was an explosion of research.
Scientists quickly applied CRISPR to study genes in plants, animals, and human cells. Researchers could disable genes to understand their functions, create disease models in laboratory animals, and explore new therapeutic strategies.
CRISPR democratized genetic engineering. What once required specialized expertise became accessible to thousands of laboratories globally.
The pace of discovery accelerated dramatically.
CRISPR and the Future of Medicine
The most exciting potential of CRISPR lies in medicine. Many diseases arise from faulty genes, making them prime targets for gene editing therapies.
Instead of treating symptoms with drugs, CRISPR offers the possibility of repairing genetic defects directly.
In genetic disorders caused by single mutations, the approach is conceptually straightforward. Scientists design CRISPR systems that locate the defective gene and correct the mutation.
Early experiments have shown promising results for conditions such as sickle cell disease. In this disorder, a mutation affects hemoglobin, the protein responsible for carrying oxygen in red blood cells.
By editing blood stem cells outside the body and returning them to patients, researchers aim to restore healthy hemoglobin production.
Other inherited diseases may eventually become treatable through similar approaches.
The dream of curing genetic diseases at their source is no longer theoretical.
Fighting Cancer with Gene Editing
Cancer arises when genetic mutations cause cells to grow uncontrollably. CRISPR offers powerful tools for studying and potentially treating these diseases.
Researchers use CRISPR to identify genes involved in cancer development and resistance to therapy. By editing these genes in laboratory models, scientists can uncover the molecular pathways that drive tumor growth.
CRISPR also enables innovative therapeutic strategies.
One approach involves modifying immune cells so they can better recognize and attack cancer cells. These engineered immune cells can be programmed to target specific tumor markers.
By editing immune cells using CRISPR, scientists aim to strengthen the body’s natural defenses against cancer.
Although many of these treatments remain experimental, early results suggest enormous potential.
CRISPR and Infectious Diseases
Beyond inherited disorders and cancer, CRISPR may play a role in combating infectious diseases.
Some viruses integrate their genetic material into human DNA. In theory, CRISPR could locate and remove these viral sequences from infected cells.
Researchers are exploring whether gene editing might one day eliminate persistent viral infections.
CRISPR-based diagnostic technologies are also emerging. These systems use CRISPR enzymes to detect specific genetic signatures of pathogens, allowing rapid identification of infections.
Such diagnostic tools could transform disease detection, particularly in regions with limited laboratory infrastructure.
The same technology originally discovered in bacteria may help humanity fight some of its most challenging diseases.
Gene Editing Beyond Humans
The influence of CRISPR extends beyond human medicine. Scientists are using gene editing to transform agriculture, environmental science, and biotechnology.
In crops, CRISPR allows researchers to introduce beneficial traits such as drought tolerance, disease resistance, and improved nutritional content.
Unlike traditional genetic modification, which often involves inserting genes from other species, CRISPR can make precise edits within existing plant genomes.
This precision may accelerate the development of crops capable of feeding a growing global population under changing climate conditions.
CRISPR is also being explored for controlling insect populations that spread diseases such as malaria. By modifying the genes of mosquitoes, scientists hope to reduce their ability to transmit parasites.
These applications highlight how gene editing could influence ecosystems and agriculture on a global scale.
Ethical Questions and Human Responsibility
With great scientific power comes profound ethical responsibility.
CRISPR raises questions that reach far beyond laboratories and clinics. The ability to edit genes touches on fundamental issues about human identity, fairness, and the limits of scientific intervention.
One of the most debated topics involves editing the human germline—genetic changes that can be inherited by future generations.
If scientists alter embryos in ways that affect reproductive cells, those changes could pass to descendants indefinitely. Such modifications might prevent inherited diseases, but they could also introduce unintended consequences.
There is also concern about the potential for “designer babies,” where genetic editing might be used to enhance physical traits or intelligence.
These possibilities raise deep social and moral questions. Who decides which genetic changes are acceptable? Could such technologies widen social inequalities?
Many scientists and ethicists argue that strong international guidelines are necessary to ensure responsible use of gene editing technologies.
The debate surrounding CRISPR reflects humanity’s struggle to balance innovation with caution.
Technical Challenges in Gene Editing
Despite its power, CRISPR is not perfect.
One of the main challenges involves accuracy. Although guide RNAs are designed to target specific DNA sequences, the system can sometimes cut unintended regions of the genome. These off-target edits may cause unwanted mutations.
Researchers are working to improve the precision of CRISPR systems by engineering new variants of Cas enzymes and refining guide RNA design.
Another challenge involves delivering CRISPR components into cells within the human body. Gene editing requires that the Cas protein and guide RNA reach the correct cells and operate safely.
Scientists are developing delivery systems using viruses, nanoparticles, and other molecular carriers to transport CRISPR tools into tissues.
These technical hurdles are gradually being addressed as the technology continues to evolve.
The Rapid Evolution of CRISPR Technology
Since its introduction, CRISPR has undergone rapid technological refinement.
New gene editing techniques derived from CRISPR allow scientists to make highly specific changes without cutting DNA completely.
Base editing enables the direct conversion of one DNA letter into another, correcting certain mutations with minimal disruption.
Prime editing represents another advanced method capable of performing precise genetic changes without double-stranded DNA breaks.
These innovations expand the possibilities of gene editing while improving safety and accuracy.
CRISPR has become not just a single tool but an entire platform for manipulating genetic information.
The Future of Genetic Medicine
As CRISPR technology matures, its impact on medicine could become transformative.
Doctors may one day diagnose genetic diseases early and correct them before symptoms appear. Personalized gene editing treatments could target the unique genetic profile of each patient.
Regenerative medicine may benefit as well. Scientists could repair damaged tissues or engineer replacement cells using genetic modifications.
The possibility of curing previously untreatable diseases inspires both excitement and caution.
Medical revolutions rarely occur overnight. Clinical trials, regulatory review, and long-term safety studies will shape the path forward.
Yet the momentum behind gene editing research continues to grow.
A New Chapter in Human History
CRISPR marks a turning point in humanity’s relationship with biology.
For thousands of years, humans have shaped life indirectly through agriculture, selective breeding, and medicine. Now, for the first time, we possess the tools to edit the genetic instructions of living organisms with extraordinary precision.
This power opens doors to curing diseases, improving food security, and advancing biotechnology.
At the same time, it challenges society to confront difficult ethical decisions about how such power should be used.
CRISPR does not simply represent a technological breakthrough. It represents a new chapter in the story of life on Earth.
Humanity now stands at the threshold of rewriting the genetic code itself.
Whether this power ultimately becomes one of our greatest achievements or one of our greatest responsibilities will depend on the wisdom with which we choose to use it.